Lipid monolayers at the air–liquid interface encode rich thermodynamic information, yet its extraction is often manual, subjective, and poorly reproducible. SurfFitter provides a fully automated and physically grounded analysis framework. Upload your surface pressure–area isotherm, and the software fits it using the Phase-Resolved Interfacial Surface Model (PRISM). The analysis yields phase boundaries, elastic moduli, chemical potential variations, and phase-specific surface coverage, all delivered in a structured report with corresponding plots and numerical descriptors. Our goal is to map compression isotherms into a minimal set of physically meaningful parameters with maximum accuracy and reproducibility.
Reference for citation: Paula Antelo-Riveiro, Angel Piñeiro, Rebeca García-Fandiño. PRISM: a phase-resolved surface equation of state across the full Langmuir isotherm. (Under Review)
Results will be displayed once the analysis is completed. This could take a few minutes.
Cut data range
The region highlighted in red will be used for the analysis. Use the slider to cut data at high surface pressure values to exclude the monolayer collapse regime when needed.
SurfFitter report is being generated...
PDF Preview
Your browser cannot display the PDF inline.
Open PDF ReportThe PDF report is still being generated.
View complete resultsSurfFitter report is being generated...
PDF Preview
Your browser cannot display the PDF inline.
Open PDF ReportThe PDF report is still being generated.
View complete resultsPRISM Model
SurfFitter model for the π-MMA isotherm
Paula Antelo-Riveiro, Angel Piñeiro, Rebeca García-Fandiño. PRISM: a phase-resolved surface equation of state across the full Langmuir isotherm. (Under Review)
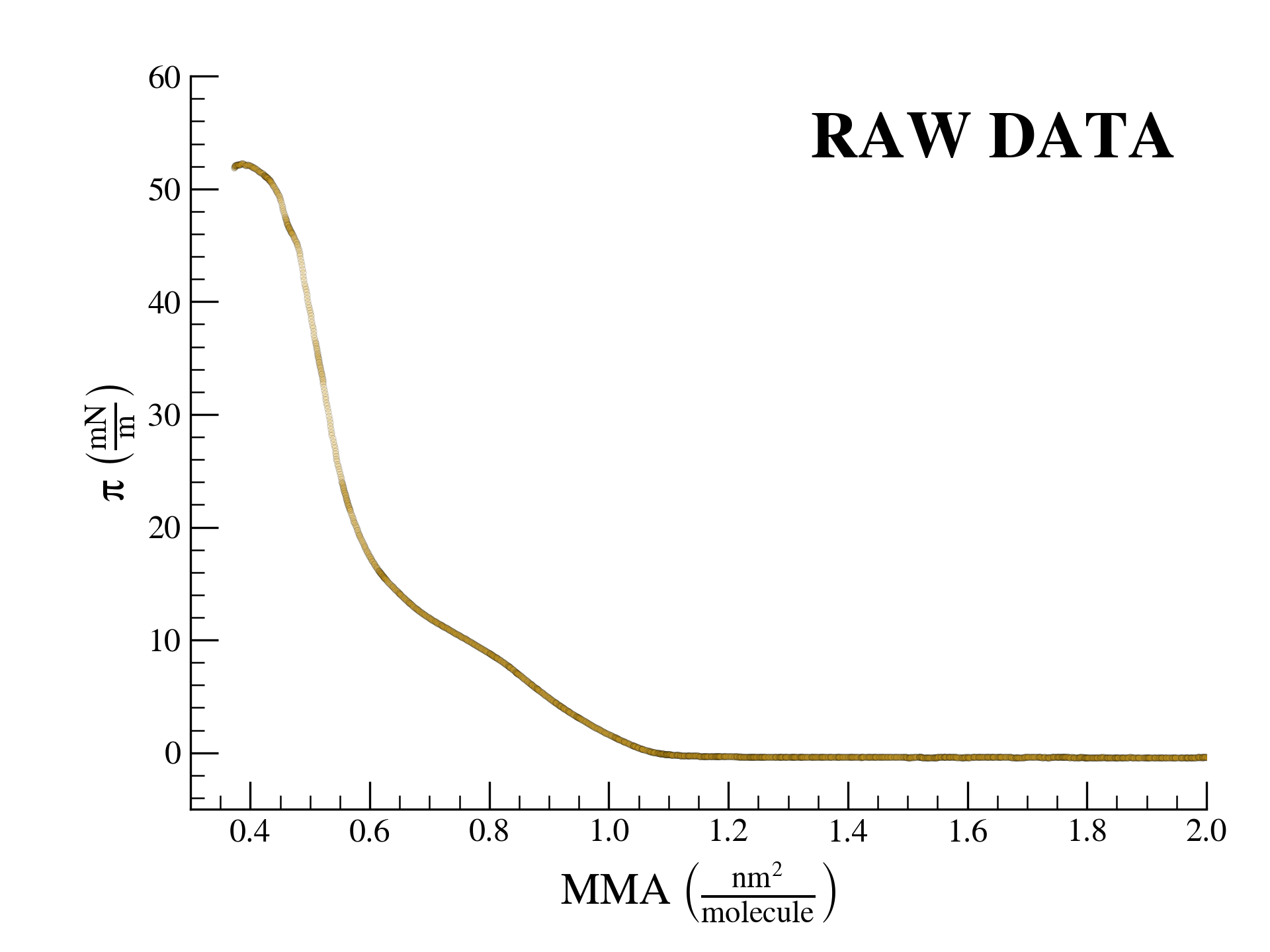
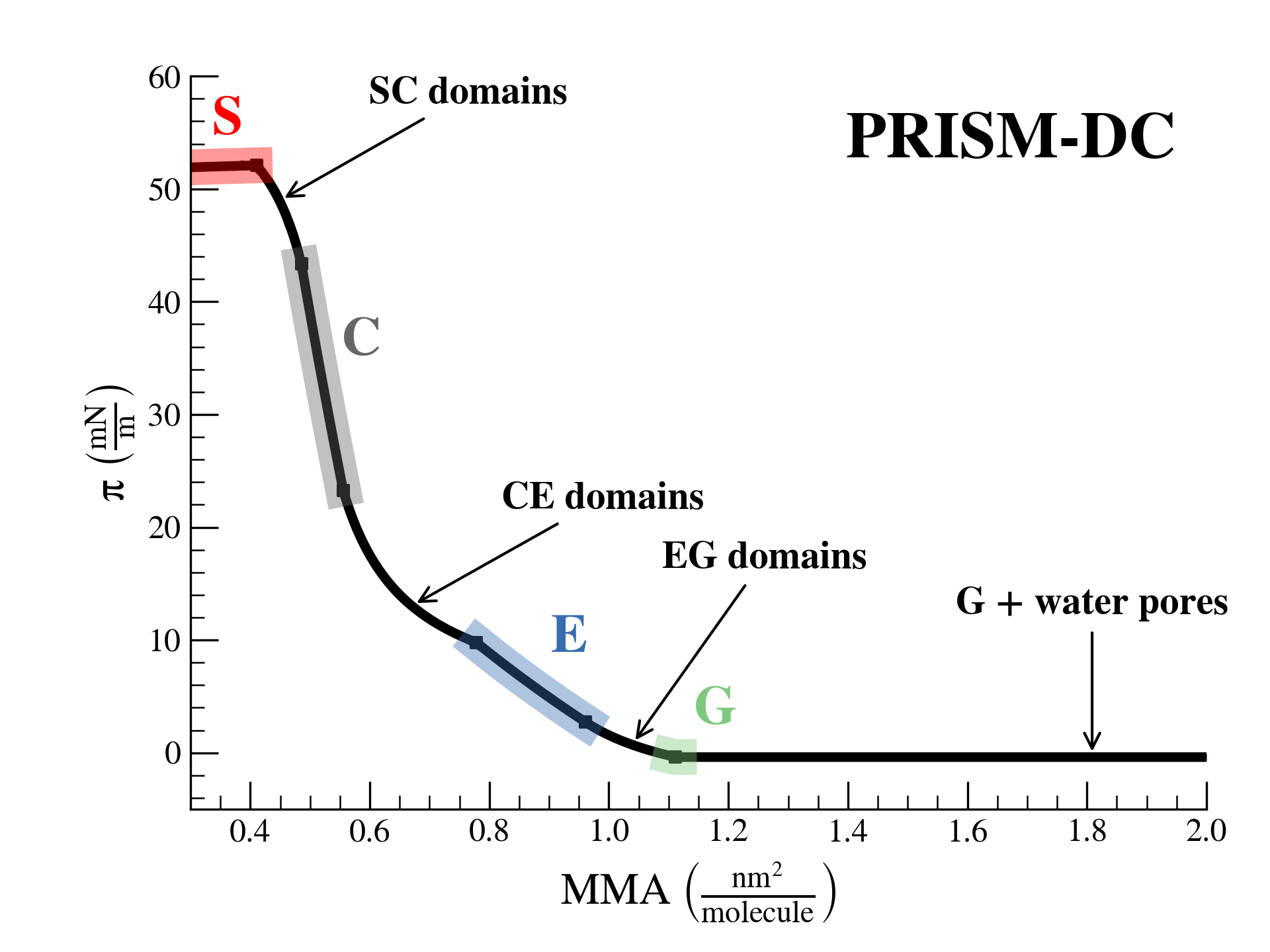
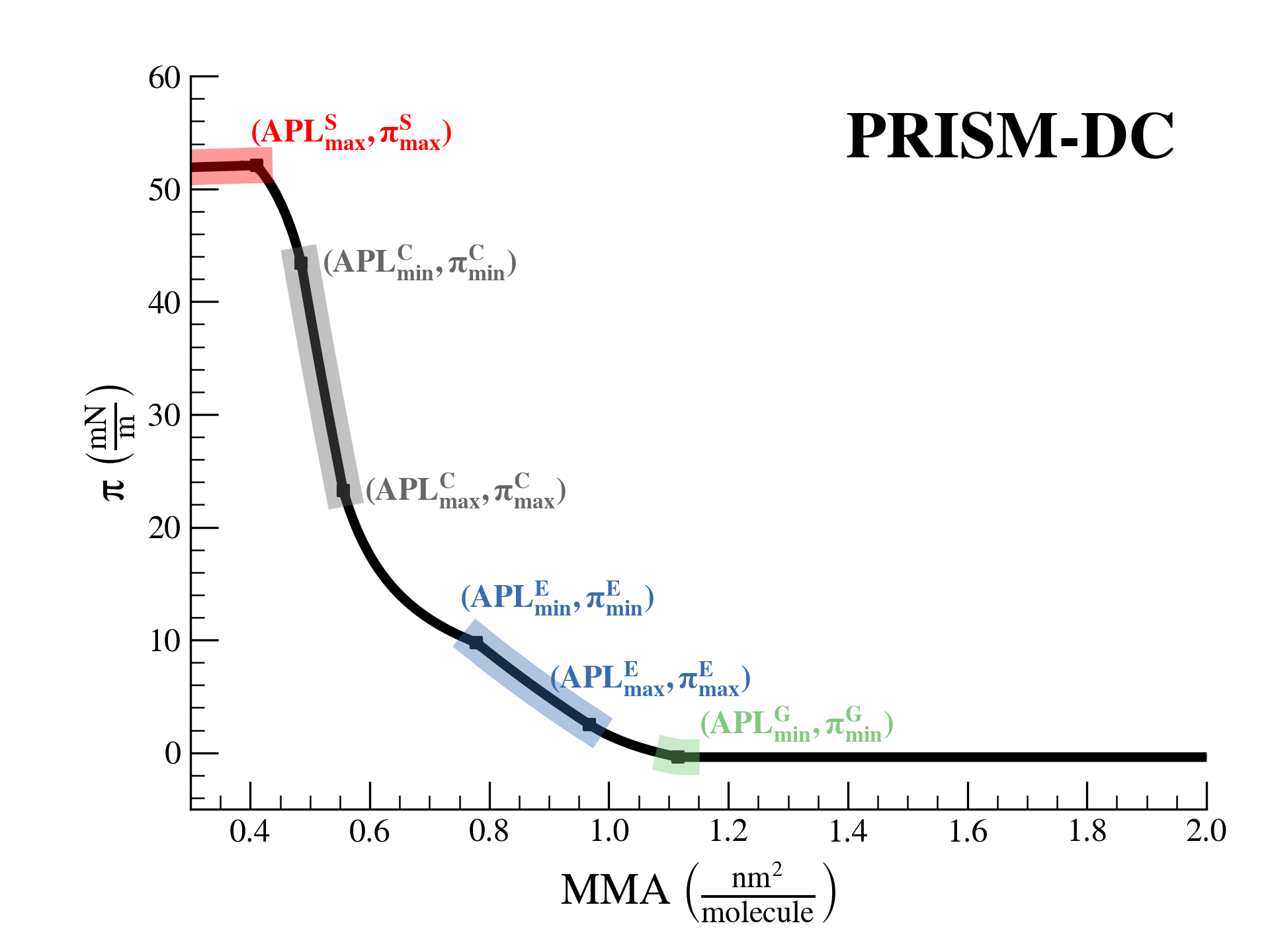
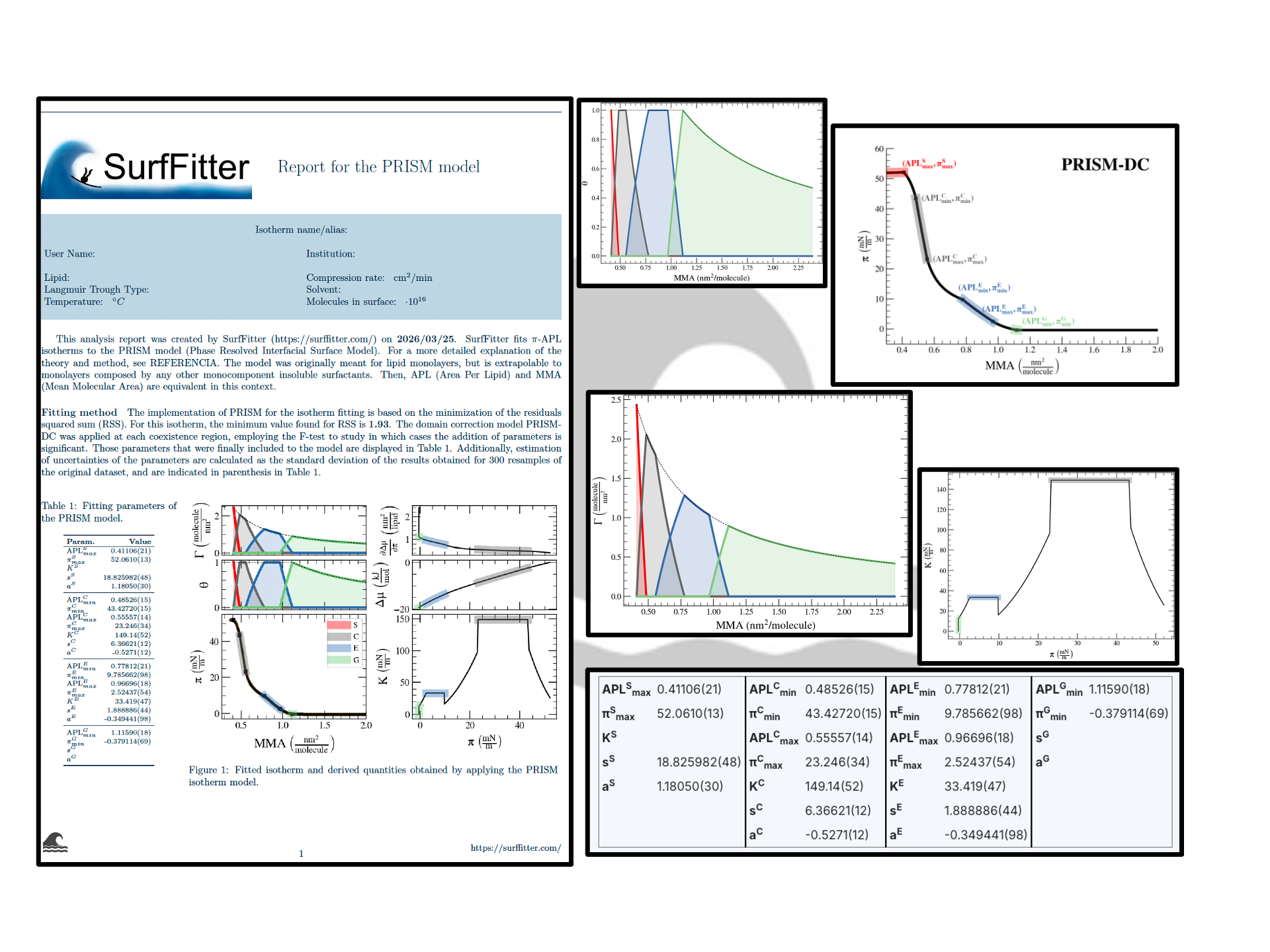
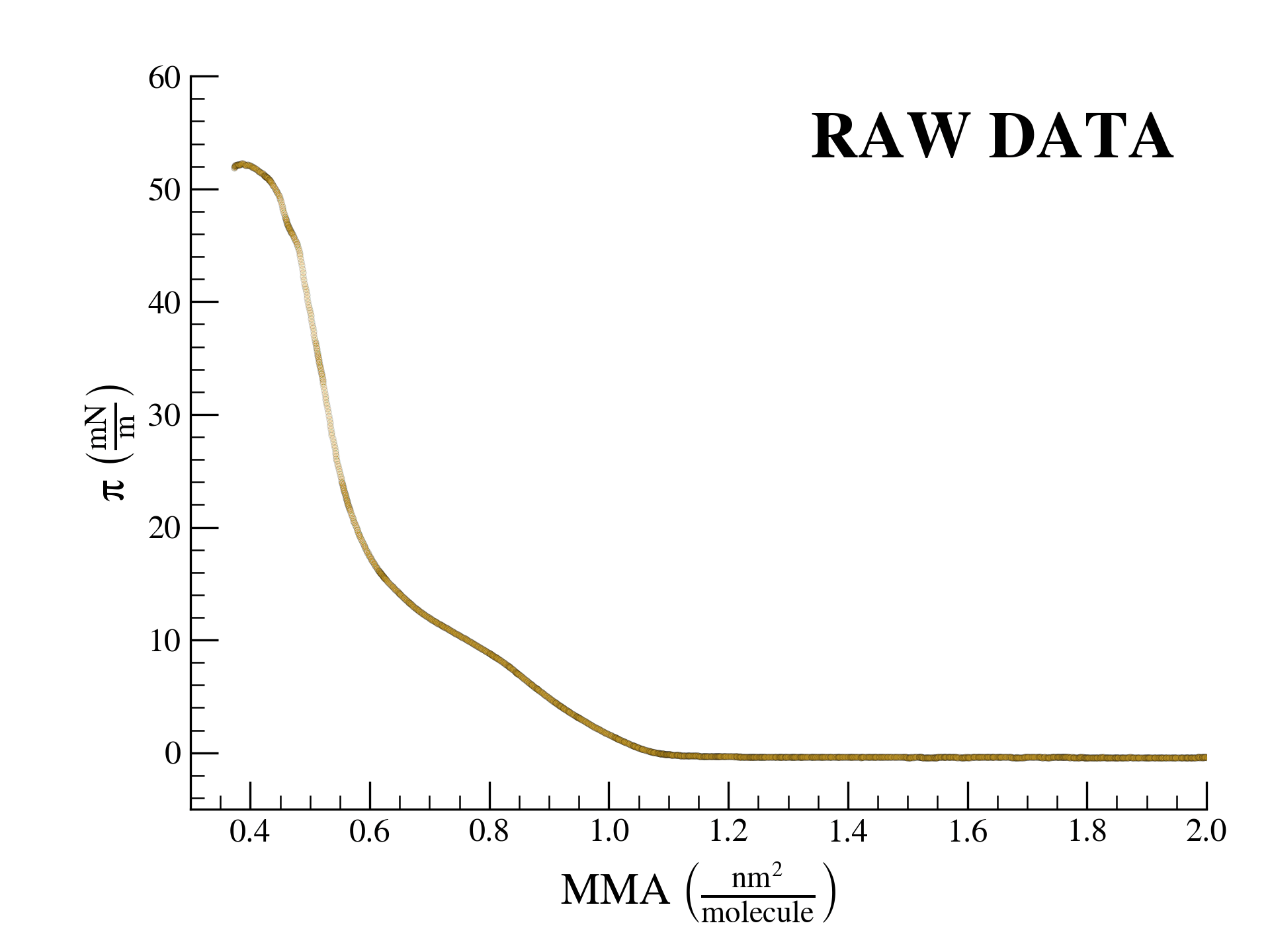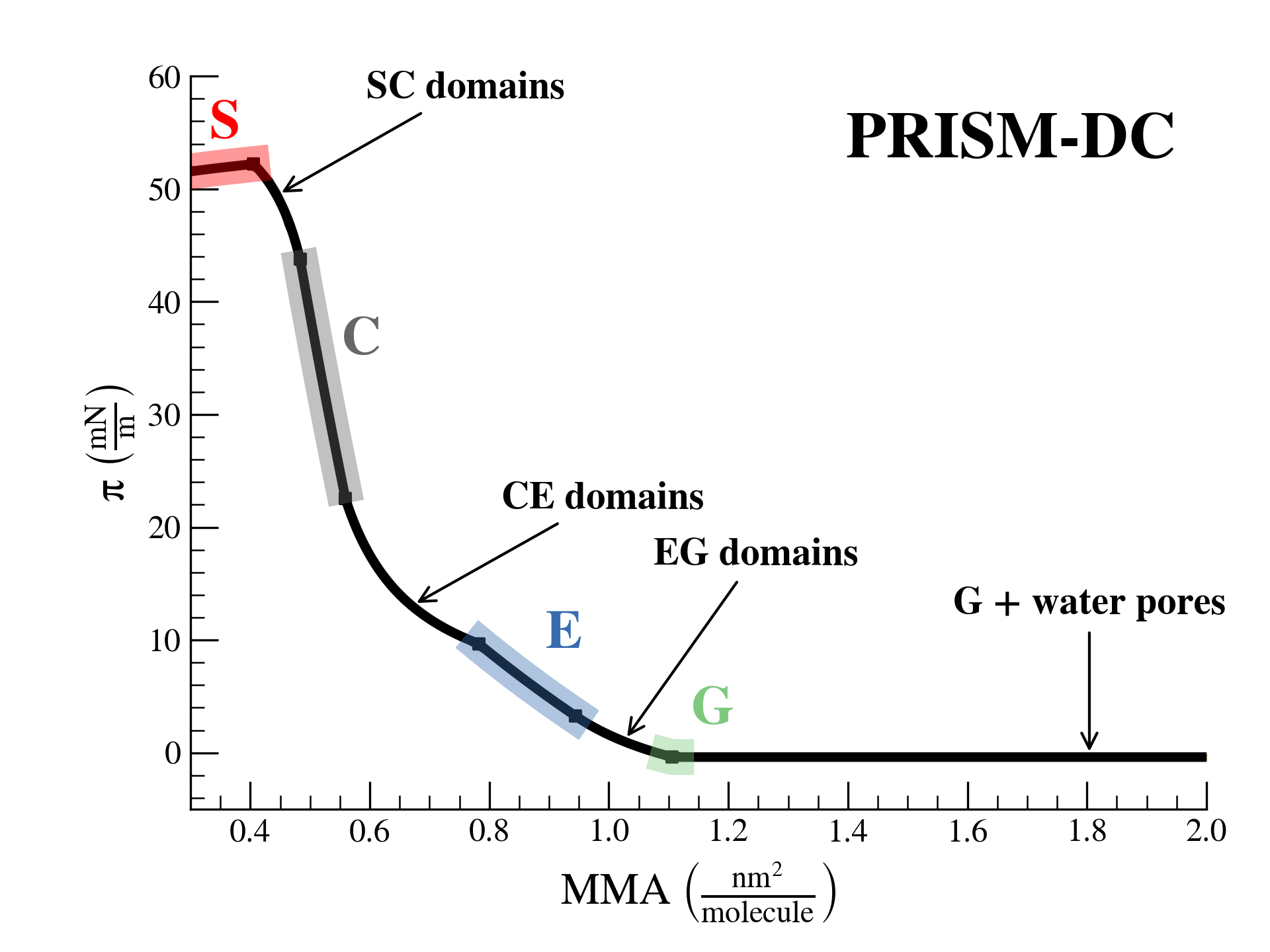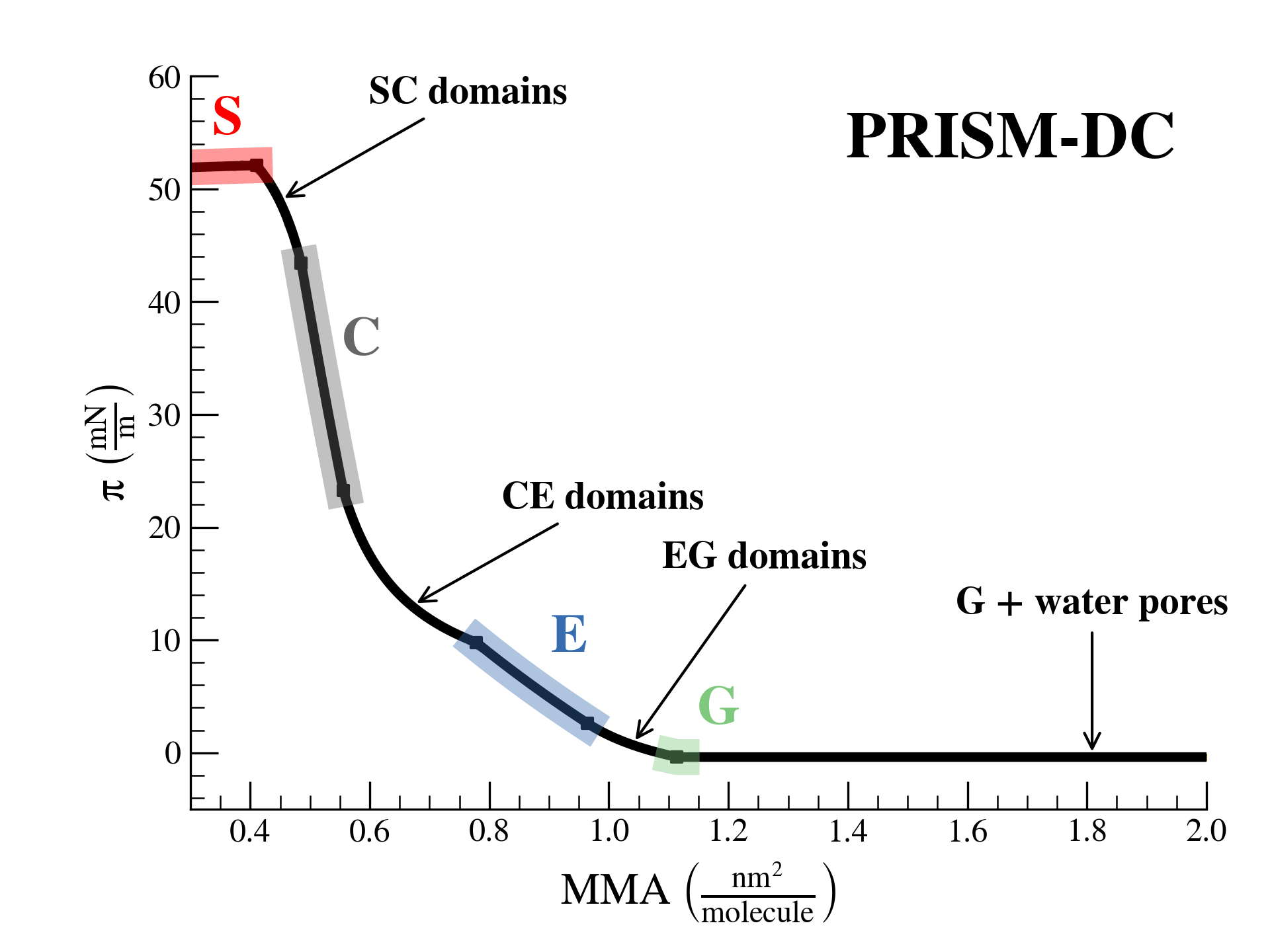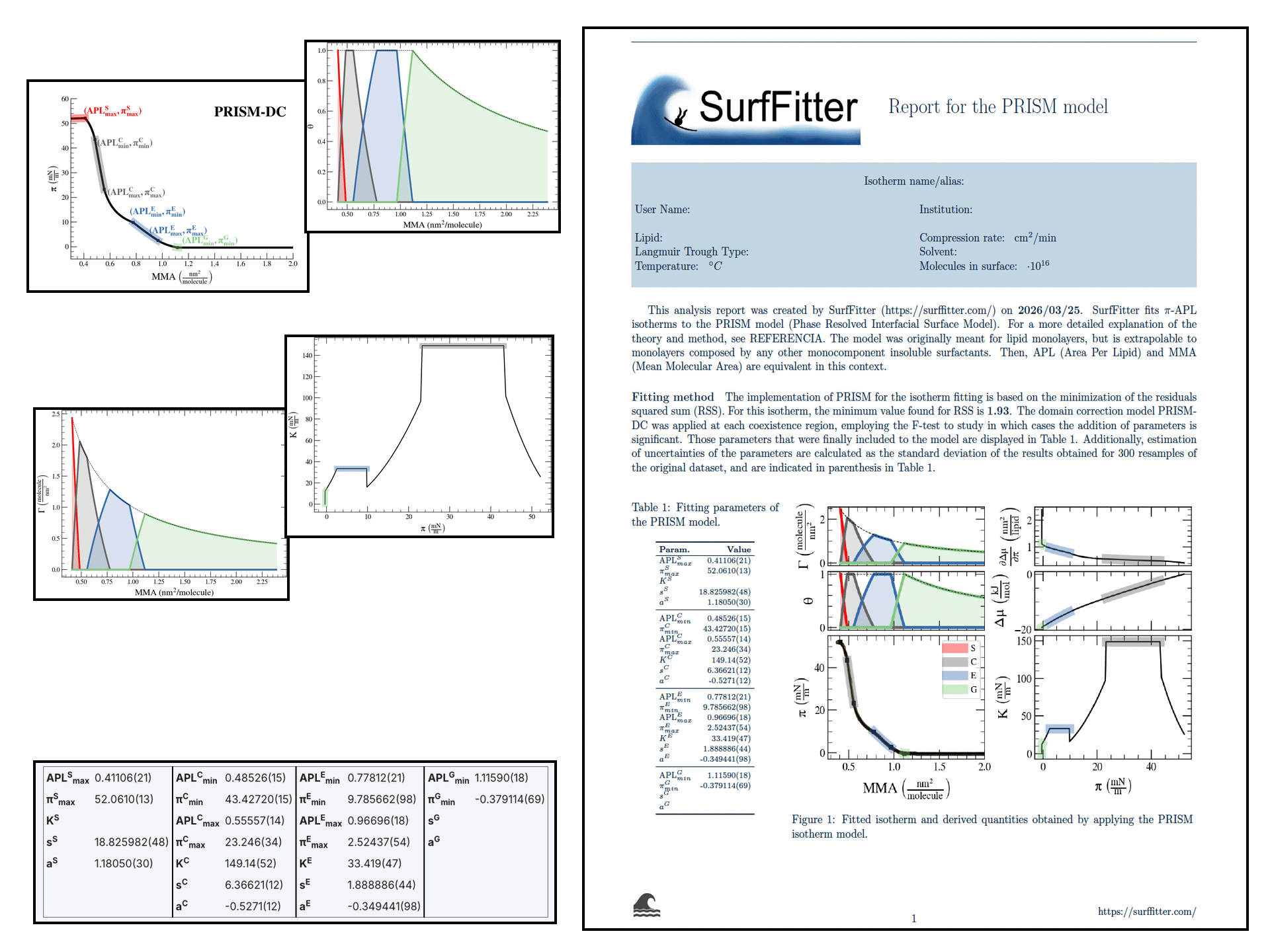
SurfFitter analyzes isotherm data applying the PRISM model, which is built on basic thermodynamic relations. The PRISM model, although initially meant for lipid monolayers, is valid for monocomponent Langmuir monolayers of any insoluble surfactant. PRISM identifies the regimes of single-phase and phase coexistence in the isotherm taking as fitting parameters all the \(\mathrm{APL}\),\(\pi\) values that mark the boundaries between phases.
- At regions of single phase \(\beta\), surfactants occupy an average molecular area between \(\mathrm{APL}^\beta_{min}\) and \(\mathrm{APL}^\beta_{max}\), and surface pressure increases with monolayer compression according to the elasticity modulus \(K^\beta\).
- At regions of coexistence of \(\beta-\beta'\) phases, the molecular area for each phase is constant across compression: \(\mathrm{APL}^\beta_{min}\) and \(\mathrm{APL}^{\beta'}_{max}\), respectively. However, the proportion of molecules in each phase evolves with \(\mathrm{APL}\). Surface pressure should remain constant at coexistence according to the Gibbs phase rule strictely (PRISM-I approach), but can the formain of phase domains might deviate the isotherm from this behavior (PRISM-DC approach). This approach introduces additional fitting parameters (\(s\) and \(a\)) that account for the line tension between phases.
Additionally, based on the isotherm initial shape SurfFitter discriminates the presence of four phases in the isotherm (S-C-E-G) or only three (C-E-G and S-C-G). In any case, the PRISM model fitted parameters allow to extract additional thermodynamic information of the system:
| Degrees of surface coverage, \(\theta^\beta\) | The ratio of surface area occupied by each phase. |
| Surface concentration, \(\Gamma^\beta\) | The number of molecules in a certain phase per unit area of the total surface. |
| Elasticity modulus, \(K\) | It is the inverse of the compressibility modulus. It is defined as \(ln\frac{d\pi}{dA}\). |
| Chemical potential, \(\Delta \mu\) | The change in chemical potential provoked by a change in surface area. |
| First derivative of the chemical potential with respect to the surface tension, \(\frac{d\Delta \mu}{d\gamma}\) | The rate of change of the chemical potential with respect to the surface tension, which is equal to the surface area. At constant temperature, \(\frac{d\Delta \mu}{d\gamma} = - \frac{d\Delta \mu}{d\pi} = A\) |
S-solid, C-condensed, E-expanded, G-gas, b-bulk water, s-surface water.
About Us
The team behind SurfFitter
SurfFitter is developed by the SIMBIOS group at University of Santiago de Compostela, in the CIQUS research center. The SIMBIOS group is focused on the study of biomolecular interactions at interfaces, with a special emphasis on lipid monolayers and bilayers. We are interested in understanding the fundamental physical principles that govern the behavior of these systems, as well as their applications in fields such as drug delivery, biosensing, and nanotechnology.

Rebeca García-Fandiño
Associate Professor at USC. She focuses on the structure, dynamics, and functional organization of lipid membranes.

Angel Piñeiro
Associate Professor at USC. His research is at the interface of computational biophysics, thermodynamics, and data science.

Paula Antelo-Riveiro
PhD candidate specializing in the thermodynamics of interfaces in lipid monolayers. The PRISM model and SurfFitter represent the core developments of her doctoral research.

Cesar Porto
Web developer specialized in full-stack development, databases, and application development.
Alternative Contact Options
For general inquiries, collaboration requests, or technical support, please reach out directly at: surffitterprism@gmail.com
